UP AND UP ACETAMINOPHEN
-
acetaminophen capsule, coated
Target Corporation
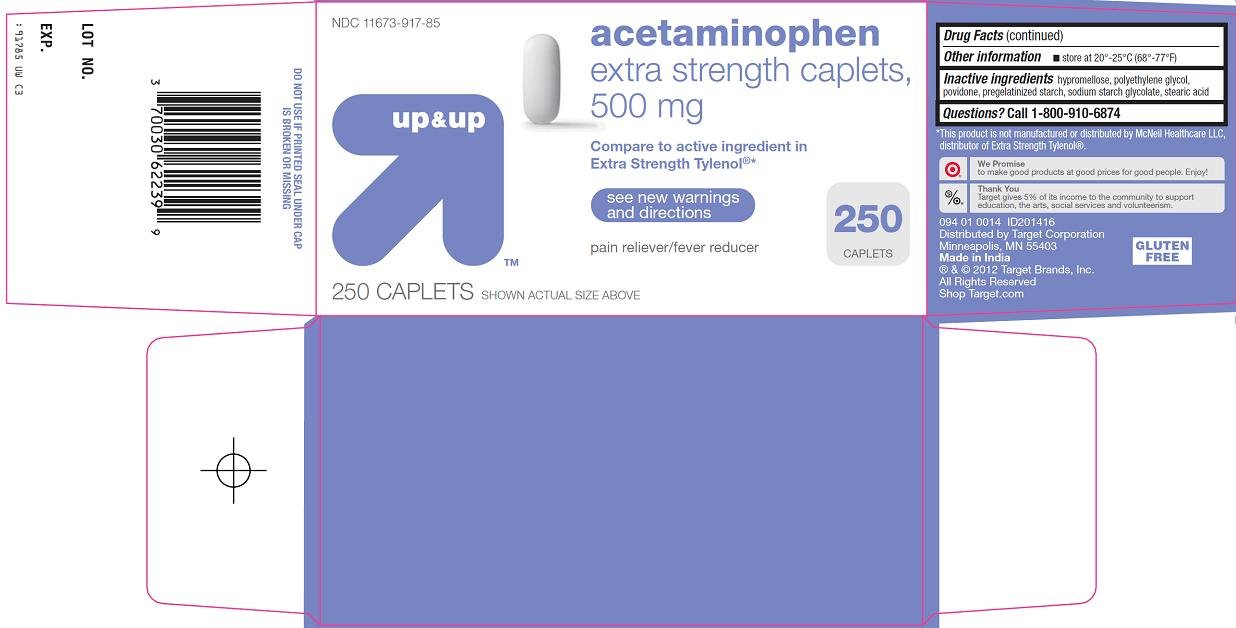
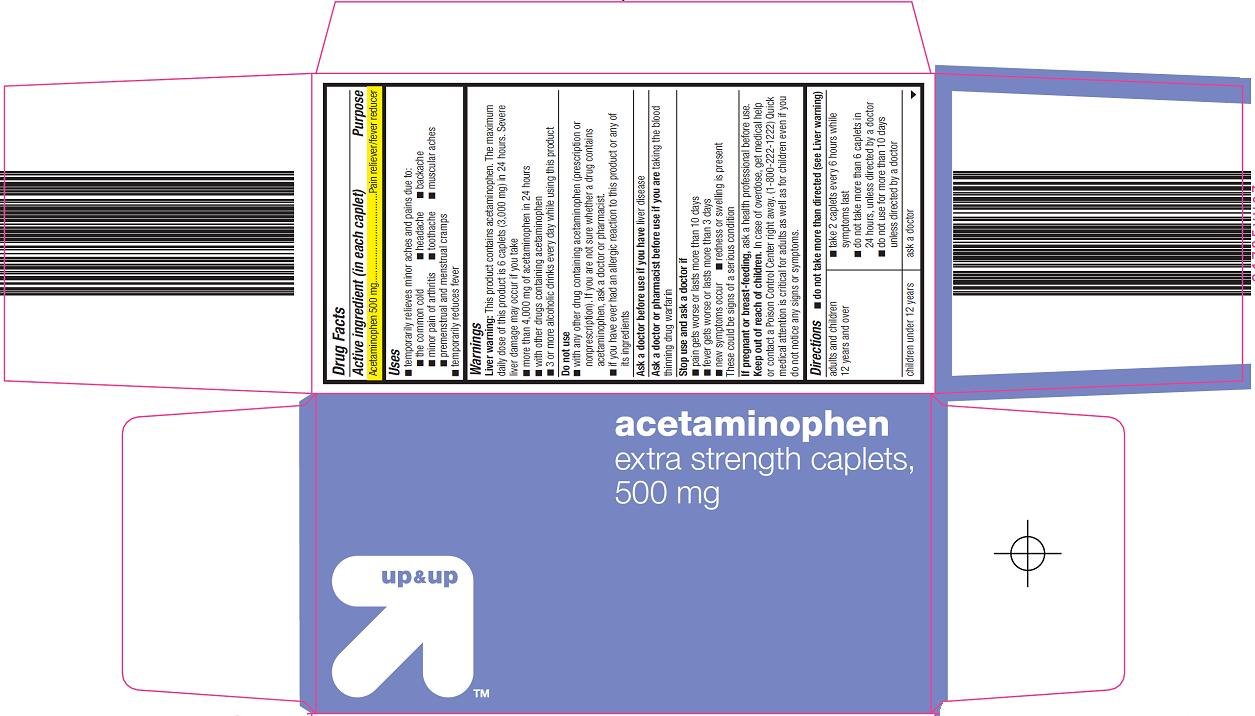
Acetaminophen 500 mg
Pain reliever/fever reducer
Liver warning: This product contains acetaminophen. The maximum daily dose of this product is 6 caplets (3,000 mg) in 24 hours. Severe liver damage may occur if you take
liver disease
taking the blood thinning drug warfarin
These could be signs of a serious condition
ask a health professional before use.
In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222) Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
|
adults and children 12 years and over |
|
| children under 12 years | ask a doctor |
hypromellose, polyethylene glycol, povidone, pregelatinized starch, sodium starch glycolate, stearic acid
Call 1-800-910-6874
acetaminophen
extra strength caplets, 500 mg
Compare to active ingredient in Extra Strength Tylenol®
see new warnings and directions
pain reliever/fever reducer
# CAPLETS (Replace # of caplets per bottle/carton with # in bottle)
SHOWN ACTUAL SIZE ABOVE
Acetaminophen Carton Image 1
Acetaminophen Carton Image 2
|
UP AND UP ACETAMINOPHEN
acetaminophen capsule, coated | ||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph not final | part343 | 07/18/2011 | |
| Labeler - Target Corporation (006961700) |