TRISEPTIN WATERLESS
-
alcohol solution
HEALTHPOINT, LTD
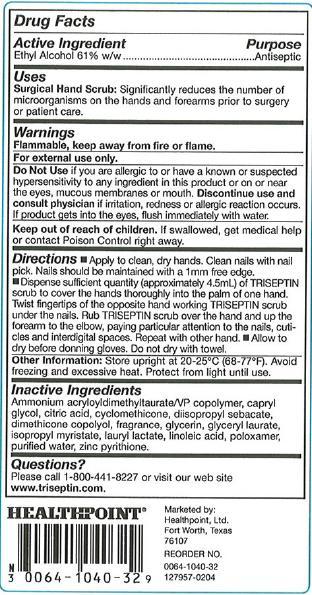
Drug Facts
Ethyl Alcohol 61% w/w
Antiseptic
Surgical Hand Scrub: Significantly reduces the number of microorganisms on the hands and forearms prior to surgery or patient care.
Flammable, keep away from fire or flame.
For external use only.
Do Not Use if you are allergic to or have a known or suspected hypersensitivity to any ingredient in this product or on or near the eyes, mucous membranes or mouth. Discontinue use and consult physician if irritation, redness or allergic reaction occurs. If product gets into the eyes, flush immediately with water.
If swallowed, get medical help or contact Poison Control right away.
Directions
Store upright at 20-25°C (68-77°F). Avoid freezing and excessive heat. Protect from light until use.
Ammonium acryloyldimethltaurate/VP copolymer, capryl glycol, citric acid, cyclomethicone, diisopropyl sebacate, dimethicone copolyol, fragrance, glycerin, glyceryl laurate, isopropyl myristate, lauryl lactate, linoleic acid, poloxamer, purified water, zinc pyrithione.
Please call 1-800-441-8227 or visit our web site www.triseptin.com.
Marketed by:
Healthpoint, Ltd.
Fort Worth, Texas
76107
Reorder No.
0064-1040-32
127957-0204
NDC 0064-1040-32
TRISEPTIN®
WATERLESS
Alcohol with Persistence
WATERLESS, BRUSH-FREE
SURGICAL SCRUB
Patented TRIZENOL™ Technology
Pat. No. 6,110,908
Directions:
Warnings:
Flammable, keep away from fire or flame. For external use only. Do Not Use if you are allergic to or have a known or suspected hypersensitivity to any ingredient in this product or on or near the eyes, mucous membranes or mouth. Discontinue use and consult physician if irritation, redness or allergic reaction occurs.
READ DRUG FACTS BEFORE USE.
HEALTHPOINT®
32 FL OZ (946 mL)
127956-0204

|
TRISEPTIN
WATERLESS ethyl alcohol solution | ||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph not final | part333E | 03/01/2023 | |
| Labeler - HEALTHPOINT, LTD (965634504) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| DPT LABORATORIES, LTD | 832224526 | MANUFACTURE | |