DRISTAN COLD MULTISYMPTOM
-
acetaminophen,
chlorpheniramine maleate and
phenylephrine hydrochloride tablet
Pfizer Consumer Healthcare
DRISTAN COLD MULTI-SYMPTOM TABLETS
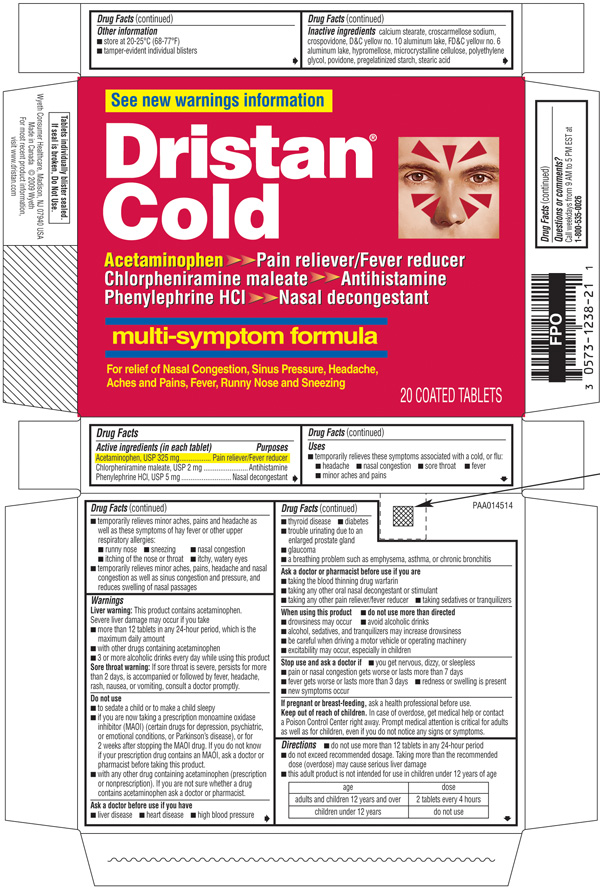
(acetaminophen, chlorpheniramine maleate, and phenylephrine HCl)ACTIVE INGREDIENTS (IN EACH TABLET)
Acetaminophen, USP 325 mg
Chlorpheniramine maleate, USP 2 mg
Phenylephrine HCl, USP 5 mg
PURPOSES
Pain reliever/Fever reducer
Antihistamine
Nasal decongestant
USES
- temporarily relieves these symptoms associated with a cold, or flu:
- headache
- nasal congestion
- sore throat
- fever
- minor aches and pains
- temporarily relieves minor aches, pains and headache as well as these symptoms of hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- nasal congestion
- itching of the nose or throat
- itchy, watery eyes
- temporarily relieves minor aches, pains, headache and nasal congestion as well as sinus congestion and pressure, and reduces swelling of nasal passages
WARNINGS
Liver warning:
This product contains acetaminophen. Severe liver damage may occur if you take
- more than 12 tablets in any 24-hour period, which is the maximum daily amount
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Sore throat warning:
if sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Do not use
- to sedate a child or to make a child sleepy
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen ask a doctor or pharmacist.
Ask a doctor before use if you have
- liver disease
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- trouble urinating due to an enlarged prostate gland
- glaucoma
- a breathing problem such as emphysema, asthma, or chronic bronchitis
Ask a doctor or pharmacist before use if you are
- taking the blood thinning drug warfarin
- taking any other oral nasal decongestant or stimulant
- taking any other pain reliever/fever reducer
- taking sedatives or tranquilizers
When using this product
- do not use more than directed
- drowsiness may occur
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
- excitability may occur, especially in children
Stop use and ask a doctor if
- you get nervous, dizzy, or sleepless
- pain or nasal congestion gets worse or lasts more than 7 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- new symptoms occur
If pregnant or breast-feeding,
ask a health professional before use.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away. Prompt medical attention is critical for adults as well as for children, even if you do not notice any signs or symptoms.
Directions
- do not use more than 12 tablets in any 24-hour period
- do not exceed recommended dosage. Taking more than the recommended dose (overdose) may cause serious liver damage
- this adult product is not intended for use in children under 12 years of age
| age |
dose |
| adults and children 12 years and over |
2 tablets every 4 hours |
| children under 12 years |
do not use |
OTHER INFORMATION
- store at 20-25°C (68-77°F)
- tamper-evident individual blisters
INACTIVE INGREDIENTS
calcium stearate, croscarmellose sodium, crospovidone, D&C yellow no. 10 aluminum lake, FD&C yellow no. 6 aluminum lake, hypromellose, microcrystalline cellulose, polyethylene glycol, povidone, pregelatinized starch, stearic acid
QUESTIONS OR COMMENTS?
Call weekdays from 9 AM to 5 PM EST at 1-800-535-0026
PRODUCT PACKAGING
The product packaging shown below represents a sample of that currently in use. Additional packaging may also be available.
See new warnings information
Dristan Cold
Acetaminophen >> Pain reliever/Fever reducer
Chlorpheniramine maleate >> Antihistamine
Phenylephrine HCl >> Nasal Decongestant
Multi-symptom formula
For relief of Nasal Congestion, Sinus Pressure, Headache, Aches and Pains, Fever, Runny Nose and Sneezing
20 COATED TABLETS
Tablets individually blister sealed. If seal is broken, Do Not Use.
Wyeth Consumer Healthcare
Madison, NJ 07940 USA
Made in Canada ©2009 Wyeth
For most recent product information, visit www.dristan.com
DRISTAN COLD MULTISYMPTOM
acetaminophen, chlorpheniramine maleate, phenylephrine hcl
tablet
|
|
|
|
|
|
Revised: 06/2010Pfizer Consumer Healthcare