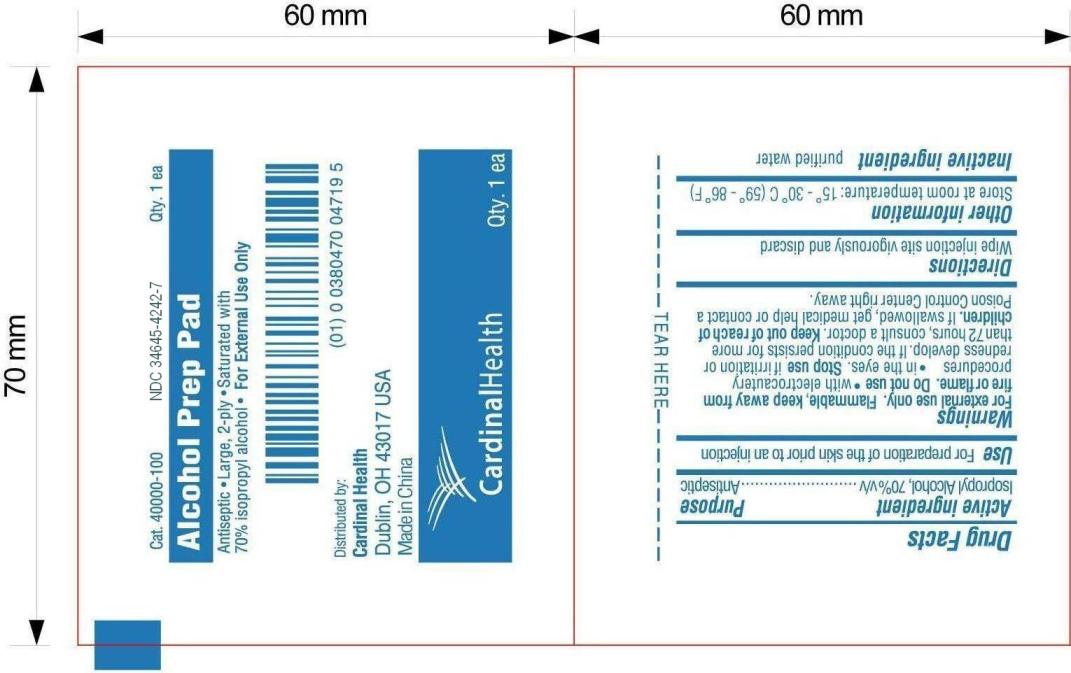
ALCOHOL PREP PAD
-
isopropyl alcohol swab
Jiangsu Province JianErKang Medical Dressing Co. ,Ltd.
Alcohol Prep
Pad
Antiseptic * Large, 2-ply * Saturated with 70% isopropyl alcohol
* For External Use Only
Distributed by:
Cardinal Health
Dublin, OH
43017 USA
Made in China
Active ingredient
Isopropyl Alcohol, 70% v/v
Purpose
Antiseptic
Use
For preparation of skin prior to injection
Warnings
For external use only.
Flammable, keep away from fire or flame.
Do not use
with electrocautery procedures, in the eyes.
Directions
Wipe injection site vigorously and discard
Other information
Store at room temperature: 15。- 30。C(59。-86。F)
Inactive ingredient
purified water
Image of Pouch Label
|
ALCOHOL PREP PAD
isopropyl alcohol swab | ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| ||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| OTC monograph final | part333 | 01/01/2023 | |
| Labeler - Jiangsu Province JianErKang Medical Dressing Co. ,Ltd. (530968767) |